
In Bohr ’s model, electrons move between orbits, depending on their energy levels. Bohr ’s model and the quantum atomic model changed the way we see the atom ’s nature and how electrons interact within it. /rebates/&252fnuclear-model.Successive atomic models, such as those proposed by Thomson and Rutherford, changed the way we think about the atom ’s charge, as they included electrical charges and described how these were distributed in the atom.
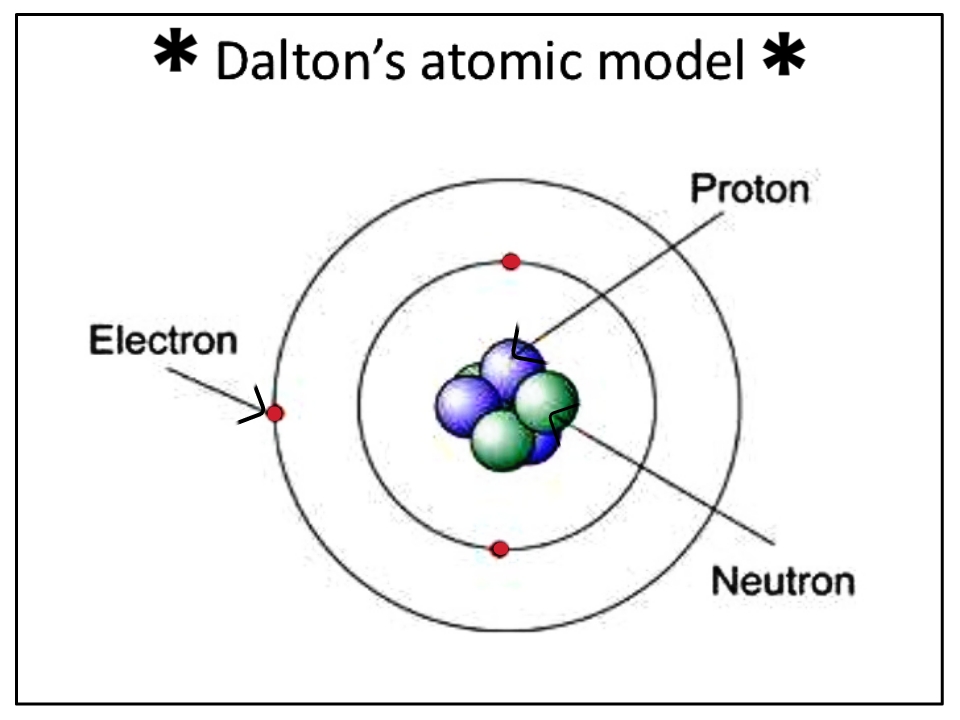
Dalton ’s model suggested that chemical reactions were the result of re-arrangements in the atoms that compose the object.He also claimed that the electrons surrounding the nucleus. Greek philosopher Democritus understood all matter to be composed of the same small objects called atoms. Rutherfords model proposed that the negatively charged electrons surround the nucleus of an atom.The atomic model has passed through various stages of development with different understandings of the structure and composition of the atom.In the model, the electrons were orbiting around the centre. Rutherford proposed that the atom contains a nucleus, with all the positive charges concentrated in the centre. The atom was almost empty inside, as not many particles fired against the foil impacted the nuclei of the atoms. However, t he experiment proved that Thomson was wrong. If the atom was a solid blob made of positive charge with some electrons on top, as Thomson ’s atomic model proposed, most of the fired particles would not reach the other side of the foil. The experiments, carried out by a student named Ernest Marsden, fired particles against a thin foil made of gold. Rutherford ’s atomic modelĪ New Zealand scientist named Ernest Rutherford designed some experiments together with German scientist Hans Geiger. Thomson ’s atomic model proposed a soup charged positively with the electrons floating on top. This model is also known as the plum pudding model.įigure 2. Thomson proposed that atoms had small negative particles floating above a fluid of positive charge. Scientists during Thomson ’s time thought that atoms were essentially neutral. Total Internal Reflection in Optical Fibre According to Rutherford model, an electron orbiting around the nucleus continuously radiates energy due to the acceleration hence the atom will not.Newton’s and Huygens’ Theories of Light.Einstein's Theory of Special Relativity.Centripetal Acceleration and Centripetal Force.Electromagnetic Radiation and Quantum Phenomena.Galileo's Leaning Tower of Pisa Experiment.Magnetic Flux and Magnetic Flux Linkage.Potential Energy and Energy Conservation.Conservative and Non Conservative Forces.
